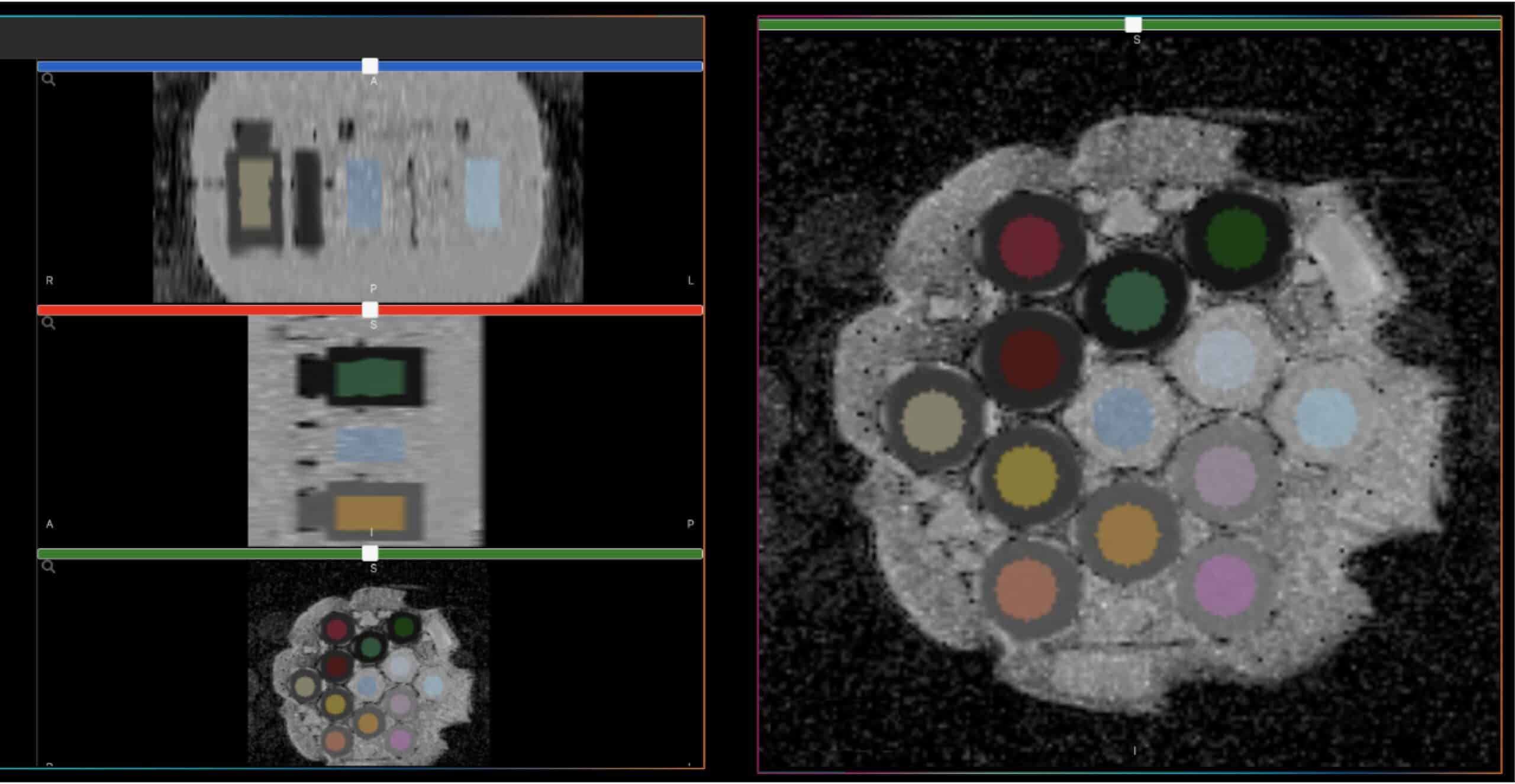
Happy Holidays Customers, Collaborators, and qMR Friends!
If you plan to attend RSNA 2023 in Chicago, IL, Nov. 26 – 30, CaliberMRI will be exhibiting at Booth 7008. Please stop by or reach out to set up a meeting to discuss standard or customer phantom needs, QA/QC, longitudinal and multi-site studies, sequence development, AI/ML, clinical trials, and any other qMRI work.
Ask us how you can receive a FREE QIBA Conformance Certification!!
We also invite you to explore our rapidly growing Resource Library!
The CaliberMRI team